
Conceptual rendering of the plating and stripping reaction of the lithium metal electrode
The high reactivity of lithium metal reduces the electrolyte at its surface, thereby leading to the degradation of lithium metal battery performance. To overcome this issue, scientists have developed functional electrolytes and electrolyte additives to form a surface protective film, which impacts the safety and efficiency of lithium batteries, but this was still not efficient to prevent certain severe side reactions. In the current study, researchers stabilized the lithium metal and electrolyte by designing the electrolyte providing upshifted oxidation-reduction potential of lithium metal, thus succeeding in weakening the reaction activity of lithium metal thermodynamically, which could help achieve better battery performance. ©2022 Yamada & Kitada Lab., Department of Chemical System Engineering, The University of Tokyo
A team of researchers has discovered a new mechanism to stabilize the lithium metal electrode and electrolyte in lithium metal batteries. This new mechanism, which does not depend on the traditional kinetic approach, has potential to greatly enhance the energy density — the amount of energy stored relative to the weight or volume — of batteries.
The team published their findings in the journal Nature Energy.
Lithium metal batteries are a promising technology with potential to meet the demands for high-energy-density storage systems. However, because of the unceasing electrolyte decomposition in these batteries, their Coulombic efficiency is low. The Coulombic efficiency, also called the current efficiency, describes the efficiency by which electrons are transferred in the battery. So a battery with a high Coulombic efficiency has a longer battery cycle life.
“This is the first paper to propose electrode potential and related structural features as metrics for designing lithium-metal battery electrolytes, which are extracted by introducing data science combined with computational calculations. Based on our findings, several electrolytes, which enable high Coulombic efficiency, have been easily developed,” said Atsuo Yamada, a professor in the Department of Chemical System Engineering at the University of Tokyo. The team’s work has the potential to provide new opportunities in the design of next-generation electrolytes for lithium metal batteries.
In lithium-ion batteries, the lithium ion moves from the positive electrode to the negative electrode through the electrolyte during charge and back when discharging. By introducing high-energy-density electrodes, the battery’s energy density can be improved. In this context, many studies have been conducted over the past decades to change the graphite negative electrode to lithium metal. However, the lithium metal has a high reactivity, which reduces the electrolyte at its surface. Because of this, the lithium metal electrode shows a poor Coulombic efficiency.
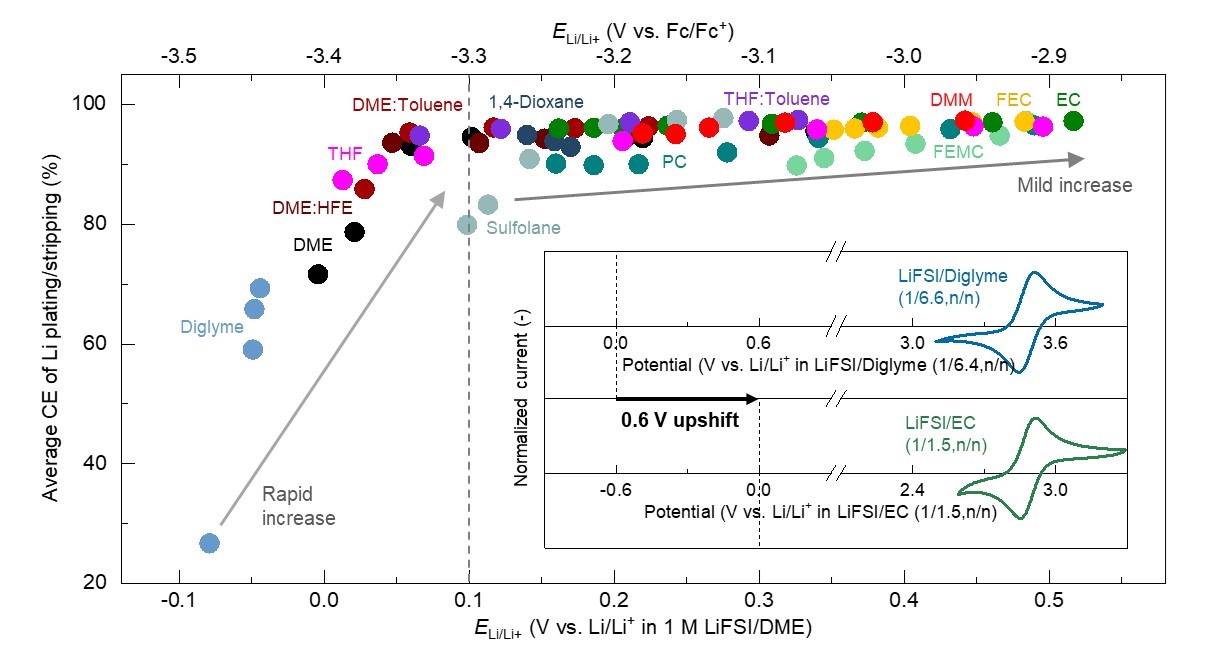
Correlation between oxidation-reduction potential of lithium metal and Coulombic efficiency
The enhanced Coulombic efficiency (CE, vertical axis), can be obtained with upshifted oxidation-reduction potential of lithium metal (ELi/Li+, horizontal axis), which lowers thermodynamic driving force to reduce the electrolyte at the lithium metal surface. The inset represents oxidation-reduction curves of the compound ferrocene (Fc/Fc+), introduced to estimate the variation of the oxidation-reduction potential of lithium metal in the given electrolytes. By comparing the oxidation-reduction potential of lithium metal in 74 different electrolytes, researchers observed a correlation between the oxidation-reduction potential and Coulombic efficiency. Based on these findings, several electrolytes, which enable high Coulombic efficiency (as high as 99.4%), have been easily developed. ©2022 Yamada & Kitada Lab., Department of Chemical System Engineering, The University of Tokyo
To overcome this problem, scientists have developed functional electrolytes and electrolyte additives that form a surface protective film. This solid electrolyte interphase has an impact on the safety and efficiency of lithium batteries. The surface protective film prevents direct contact between the electrolyte and lithium metal electrode, thereby kinetically slowing the electrolyte reduction. Yet, until now, scientists had not fully understood the correlation between the solid electrolyte interphase and the Coulombic efficiency.
Scientists know that if they improve the stability of the solid electrolyte interphase, then they can slow the electrolyte decomposition and the battery’s Coulombic efficiency is increased. But even with advanced technologies, scientists find it difficult to analyze the solid electrolyte interphase chemistry directly. Most of the studies about the solid electrolyte interphase have been conducted with indirect methodologies. These studies provide indirect evidence, therefore making it hard to develop the electrolyte stabilizing lithium metal that leads to a high Coulombic efficiency.
The research team determined that if they could upshift the oxidation-reduction potential of the lithium metal in a specific electrolyte system, they could decrease the thermodynamic driving force to reduce the electrolyte, and thus achieve a higher Coulombic efficiency. This strategy had rarely been applied in developing batteries with lithium metal. “The thermodynamic oxidation-reduction potential of lithium metal, which varies significantly depending on the electrolytes, is a simple yet overlooked factor that influences the lithium metal battery performance,” said Atsuo Yamada.
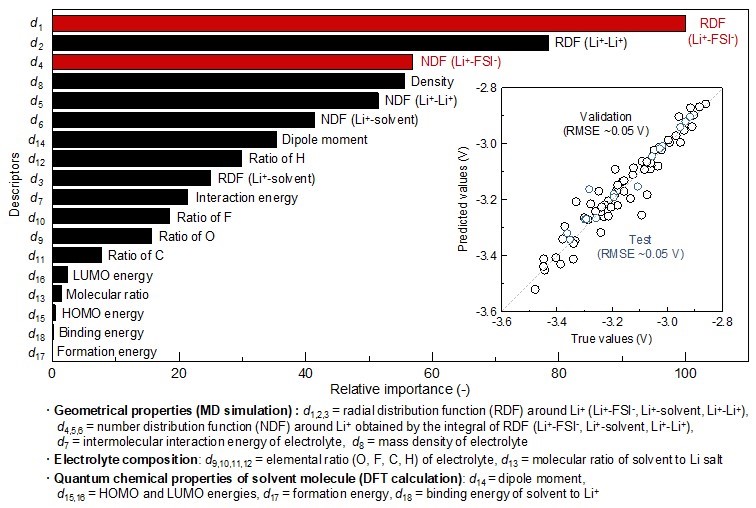
Mechanism behind the variation of oxidation-reduction potential of lithium metal revealed by introducing data science combined with computational calculations
The relative importance of descriptors for the oxidation-reduction potential of lithium metal was obtained from partial least square (PLS) regression analysis. The correlation between the predicted and observed true values of the oxidation-reduction potential of lithium metal is well fitted, which is shown as an inset figure, along with the root mean squared error (RMSE). A numerous data related to the solution structure and physicochemical properties of electrolytes were collected by MD and DFT computational calculations, and their effect to the oxidation-reduction potential of lithium metal has been quantitatively analyzed with machine learning-based regression analysis. A specific factor, coordination state of Li+ and anion FSI–, has been revealed as a most important descriptor to determining the oxidation-reduction potential of lithium metal. ©2022 Yamada & Kitada Lab., Department of Chemical System Engineering, The University of Tokyo
The team studied the oxidation-reduction potential of lithium metal in 74 types of electrolytes. The researchers introduced a compound called ferrocene into all the electrolytes as an IUPAC (International Union of Pure and Applied Chemistry)-recommended internal standard for electrode potentials. The team proved that there is a correlation between the oxidation-reduction potential of lithium metal and the Coulombic efficiency. They obtained the high Coulombic efficiency with the upshifted oxidation-reduction potential of lithium metal.
Looking ahead to future work, the research team’s goal is to unveil the rational mechanism behind the oxidation-reduction potential shift in more detail. “We will design the electrolyte guaranteeing a Coulombic efficiency of greater than 99.95%. The Coulombic efficiency of lithium metal is less than 99%, even with advanced electrolytes. However, at least 99.95% is required for the commercialization of lithium metal-based batteries,” said Atsuo Yamada.
This study was carried out in collaboration with the Nagoya Institute of Technology.







